|
An example is Ba(NO 3) 2.Trending Questions What is carbon tetra fluoride used for? Earth travels in an orbit as it revolves earths axis is always tilted in the same direction? A gas at the same temperature as it's liquid state? What is the chemical reaction that causes an egg to become hard-boiled? Would vapor pressure be higher in warm air or cold air? Indicate the most common charge of elements sulfur? What is the difference between streams and springs? Can you recycle mahogany? Is Mt. Atomic Number - Each atom has an atomic number. It has 35 isotopes with mass numbers ranging from 94 to 128 of which only Ag-107 and Ag-109 have a stable half-life. 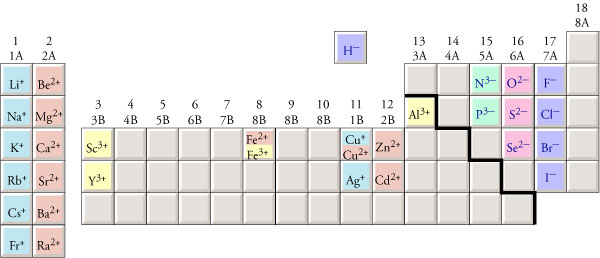
Though it is not highly reactive, it gradually tarnishes in air to form a black coating of silver sulfide. Many of the transition metals (orange) can have more than one charge. The alkaline earth metals (red) always form +2 ions. Remember that the balanced equation's coefficients state the stoichiometric factor or mole ratio of reactants and products. The alkali metals (shown in yellow) always form +1 ions. The balanced equation must now be used to convert moles of Fe (s) to moles of H 2 (g). Each elements symbol is composed of a capital letter followed by one or two lowercase letters. Silvers element symbol Ag, is from the Latin word argentum meaning silver. Silver (pronounced as SIL-ver) is a shiny metal denoted by the chemical symbol Ag. Many of the elements on the periodic table will always form ions that have the same charge. The Germans tried very hard during the second World War but failed miserably. Sea water contains around 3 parts in a billion of gold, but there's never been found an economic means of recovering it. 28 other isotopes can be produced artificially and are all radioactive. 
Determine the number of protons and electrons in an atom. 100 of gold found naturally is isotope Au-197. For example, silicon has a chemical symbol 'Si'. Describe the locations, charges, and masses of the three main subatomic particles. The number of protons in the nucleus is called the atomic number (Z) and is the property that defines an atom’s elemental identity. Electrons are light particles with a charge of 1 and a mass of 0.00055 amu. It is one of the least reactive chemical elements and is solid under standard conditions. Chemically, gold is a transition metal and a group 11 element. For example, Cu(NO 3) 2 is copper(II) nitrate, because the charge of two nitrate ions (NO 3) is 2 × 1 2, and since the net charge of the ionic compound must be zero, the Cu ion has a 2+ charge. It is a bright, slightly orange-yellow, dense, soft, malleable, and ductile metal in pure form. For cations that take on multiple charges, the charge is written using Roman numerals in parentheses immediately following the element name. This symbol usually originates from its name or its Latin name. Neutrons are relatively heavy particles with no charge and a mass of 1.0087 amu. Gold is a chemical element with the symbol Au (from Latin aurum 'gold') and atomic number 79. 
This is to show that the subscript applies to the entire polyatomic ion. Symbol - Each element is assigned a chemical symbol. For silver the preeminently important oxidation state in all of its ordinary chemistry is the state +1, although the states +2 and +3 are known. If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
